Drug intermediates list are the crucial building blocks in the pharmaceutical industry, representing compounds formed during the synthesis of an Active Pharmaceutical Ingredient (API). Their quality, availability, and cost directly impact the final drug product's efficacy and accessibility. Understanding the intricacies of drug intermediates list—from sourcing and manufacturing to quality control—is paramount for pharmaceutical companies, regulatory bodies, and ultimately, patient health globally. This knowledge is vital for ensuring a stable and secure supply chain of life-saving medications.
The global market for drug intermediates list is experiencing significant growth, driven by factors such as increasing demand for generic drugs, rising prevalence of chronic diseases, and expanding pharmaceutical research and development. According to reports, the market is projected to reach substantial figures in the coming years, highlighting the economic importance of this sector. However, this growth isn't without challenges. Issues like supply chain disruptions, regulatory scrutiny, and the need for sustainable manufacturing practices demand a proactive and informed approach to managing drug intermediates list effectively.
Navigating the complexities of drug intermediates list requires a comprehensive understanding of chemical synthesis, quality assurance, and global regulations. A robust strategy for sourcing, manufacturing, and managing these compounds is essential for pharmaceutical companies to maintain a competitive edge and deliver safe, effective, and affordable medications to patients worldwide. Investing in innovative technologies and establishing strong partnerships are key to overcoming current challenges and capitalizing on future opportunities within this dynamic landscape.

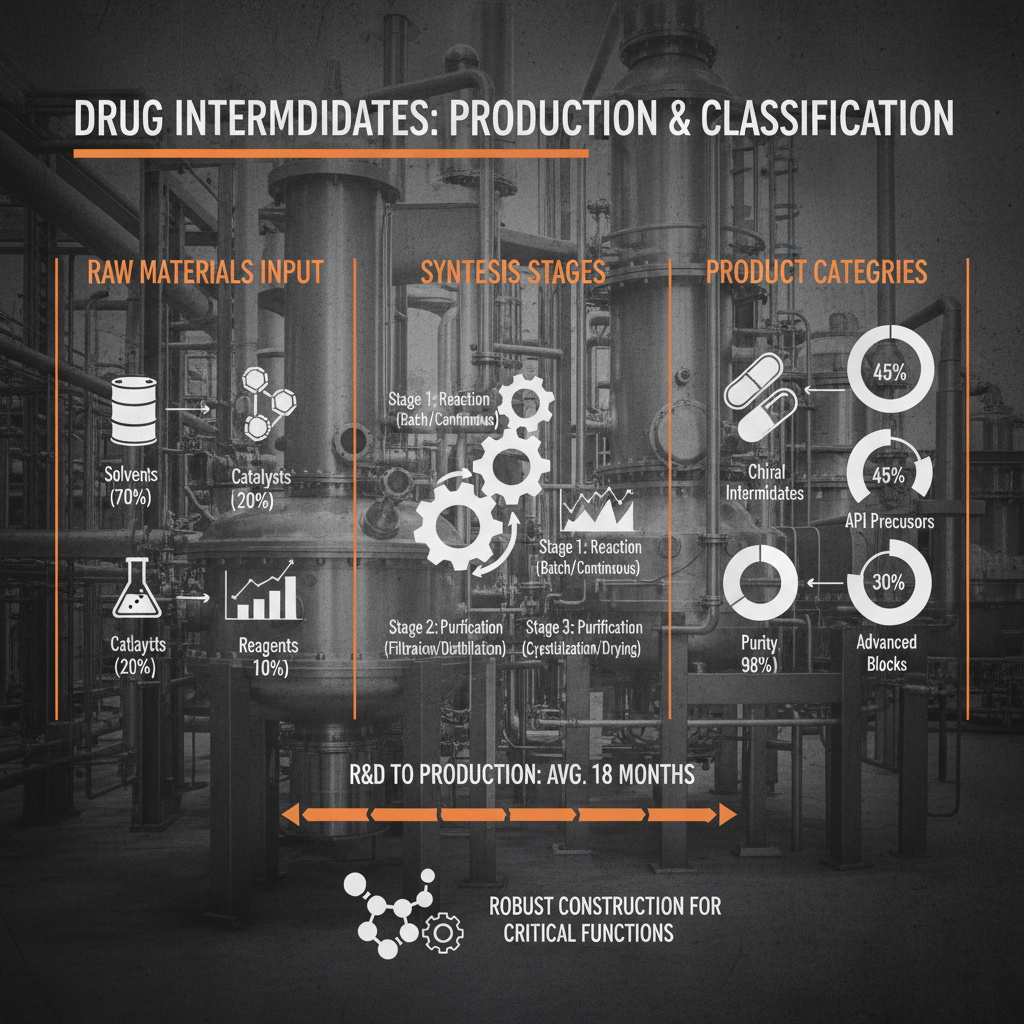
The pharmaceutical industry's reliance on a consistent and reliable supply of drug intermediates list is undeniable. Global events, such as geopolitical instability and pandemics, have highlighted the vulnerabilities within these supply chains. The concentration of manufacturing in specific regions—particularly Asia—creates a potential single point of failure. According to the United Nations Conference on Trade and Development (UNCTAD), diversifying sourcing and strengthening regional manufacturing capabilities are crucial steps toward building more resilient pharmaceutical supply chains. This drive necessitates a thorough understanding of the various players involved in the drug intermediates list market, from raw material suppliers to finished product manufacturers.
The increasing demand for complex generic drugs is also driving innovation within the drug intermediates list sector. These medications often require more intricate synthetic pathways and higher-quality intermediates. Consequently, manufacturers are investing in advanced technologies, such as continuous flow chemistry and biocatalysis, to improve efficiency and reduce costs. The International Council for Harmonisation (ICH) guidelines play a critical role in ensuring the quality and safety of these intermediates, setting standards for impurity profiling and process validation. Compliance with these regulations is essential for accessing global markets and maintaining consumer trust.
Drug intermediates list are chemical compounds that are formed during the manufacturing process of an Active Pharmaceutical Ingredient (API) but are not themselves the API. They are essentially the building blocks—the stepping stones—in the complex chemical synthesis required to create the final drug. Their structure is closely related to the API, but they lack the specific pharmacological activity. Identifying and characterizing these intermediates is a crucial part of ensuring the purity and safety of the final medication.
The importance of drug intermediates list extends beyond their chemical properties. They represent a critical link between raw materials and the finished drug product. Managing these intermediates effectively requires careful attention to process control, quality assurance, and regulatory compliance. In modern pharmaceutical manufacturing, the efficient production of high-quality drug intermediates list is vital for reducing costs, shortening development timelines, and ultimately, making life-saving medications more accessible to patients globally.
Understanding the definition and purpose of drug intermediates list is particularly important in the context of generic drug manufacturing. Generic drug companies often rely on alternative synthetic pathways to produce APIs, which can involve different intermediates compared to the original innovator drug. Therefore, thorough analytical testing and characterization are essential to demonstrate the equivalence of the generic drug to the branded version.
Several key factors determine the quality, cost, and reliability of drug intermediates list. First and foremost is Purity. High purity intermediates are essential to prevent the formation of unwanted byproducts and ensure the safety and efficacy of the final API. Stringent analytical methods, such as HPLC and NMR, are employed to verify purity levels. Another crucial aspect is Scalability. The manufacturing process for an intermediate must be scalable to meet the demands of commercial production, which can range from kilograms to tons.
Cost-Effectiveness plays a significant role, particularly in the competitive generic drug market. Optimizing synthetic routes, utilizing readily available raw materials, and minimizing waste are all strategies for reducing production costs. Furthermore, Regulatory Compliance is non-negotiable. Intermediates must be manufactured in accordance with Good Manufacturing Practices (GMP) and meet the stringent requirements of regulatory agencies like the FDA and EMA. Finally, Supply Chain Security is paramount. Ensuring a stable and reliable supply of intermediates requires diversifying sourcing, establishing strong relationships with suppliers, and implementing robust risk management strategies.
These core components are interconnected. For example, achieving high purity often comes at a cost, requiring more sophisticated purification techniques. Similarly, scaling up a process can introduce new challenges related to process control and impurity formation. Therefore, a holistic approach that considers all these factors is crucial for successful drug intermediates list manufacturing.
Drug intermediates list are used extensively in the manufacture of a wide range of pharmaceuticals, from small-molecule drugs to complex biologics. In developed countries like the United States and Europe, these intermediates are primarily used by large pharmaceutical companies and generic drug manufacturers. However, in emerging economies like India and China, a significant portion of the drug intermediates list production is geared towards export, serving as key suppliers to the global pharmaceutical industry.
Consider the production of antibiotics: numerous drug intermediates list are needed to synthesize various beta-lactam and macrolide antibiotics, forming a complex network of chemical reactions. Similarly, the production of anti-cancer drugs often involves highly specialized and complex intermediates, requiring advanced manufacturing capabilities. In remote industrial zones, particularly in Asia, these facilities often operate under stringent environmental and safety regulations.
Beyond traditional pharmaceutical manufacturing, drug intermediates list also find applications in veterinary medicine, agricultural chemicals, and even material science. The versatility of these compounds makes them valuable building blocks for a diverse range of industries. They also play a vital role in the research and development of new drugs, providing researchers with the tools to explore novel chemical structures and therapeutic targets.
Investing in high-quality drug intermediates list offers significant advantages to pharmaceutical companies. Cost savings are a primary benefit, as efficient synthesis and optimized processes reduce overall manufacturing expenses. Improved product quality and reduced impurity levels enhance patient safety and increase brand reputation. Furthermore, a reliable supply chain of drug intermediates list minimizes the risk of disruptions and ensures consistent product availability.
The long-term value extends beyond economic considerations. Sustainable manufacturing practices, such as waste reduction and the use of environmentally friendly solvents, contribute to a more responsible and ethical pharmaceutical industry. This enhances public trust and demonstrates a commitment to corporate social responsibility. The stability and predictability of drug intermediates list sourcing foster innovation and accelerate the development of new medicines.
The future of drug intermediates list is inextricably linked to advancements in green chemistry and digital transformation. Continuous flow chemistry, which offers improved process control and scalability, is gaining traction as a more sustainable alternative to traditional batch processing. Biocatalysis, utilizing enzymes to catalyze chemical reactions, is another promising area, offering higher selectivity and reduced environmental impact.
Digital technologies, such as artificial intelligence (AI) and machine learning (ML), are being employed to optimize synthetic routes, predict reaction outcomes, and identify potential impurities. These tools can accelerate process development and improve product quality. The adoption of blockchain technology is also being explored to enhance supply chain transparency and traceability, ensuring the authenticity and integrity of drug intermediates list.
One of the primary challenges facing the drug intermediates list industry is the increasing regulatory scrutiny. Agencies like the FDA are demanding more comprehensive data on intermediate manufacturing processes and impurity profiles. Another significant challenge is the vulnerability of supply chains to disruptions, as demonstrated by recent global events. The reliance on single suppliers and geographically concentrated manufacturing hubs creates risks.
To address these challenges, pharmaceutical companies are implementing strategies such as diversifying sourcing, establishing dual manufacturing sites, and investing in advanced analytical technologies. Collaborative partnerships with suppliers and research institutions are also crucial for developing innovative solutions. Adopting a proactive approach to risk management and implementing robust quality control systems are essential for ensuring a reliable and compliant supply of drug intermediates list.
Furthermore, embracing principles of green chemistry and sustainable manufacturing is not only ethically responsible but also economically advantageous in the long run. By reducing waste, minimizing energy consumption, and utilizing renewable resources, companies can improve their environmental footprint and enhance their bottom line.
| Challenge Area | Severity (1-10) | Proposed Solution | Implementation Cost (Estimate) |
|---|---|---|---|
| Regulatory Compliance | 9 | Enhanced Quality Control Systems, Data Integrity Programs | $500,000 - $1 Million |
| Supply Chain Vulnerability | 8 | Diversified Sourcing, Dual Manufacturing Sites | $1 Million - $5 Million |
| Cost Pressure | 7 | Process Optimization, Waste Reduction | $200,000 - $500,000 |
| Environmental Impact | 6 | Green Chemistry Principles, Sustainable Manufacturing | $300,000 - $700,000 |
| Analytical Complexity | 7 | Advanced Analytical Technologies (HPLC, NMR, Mass Spectrometry) | $400,000 - $800,000 |
| Skilled Workforce Shortage | 5 | Training Programs, Collaboration with Universities | $100,000 - $300,000 |
Drug intermediates are compounds formed during the synthesis of an API, lacking the final drug's pharmacological activity. APIs are the active ingredients responsible for the therapeutic effect. Intermediates are stepping stones; APIs are the final product. Strict quality control is essential for both, but intermediates focus on process control, while APIs emphasize purity and efficacy for patient use.
Mitigation involves diversifying sourcing beyond single suppliers, especially those concentrated in specific regions. Establishing dual manufacturing sites, maintaining safety stock, and implementing robust supply chain monitoring systems are also crucial. Strong supplier qualification programs and long-term contracts with reliable partners contribute to a more resilient supply chain. Proactive risk assessments and contingency planning are key.
Green chemistry focuses on minimizing environmental impact through sustainable practices. In drug intermediates, this means using less hazardous chemicals, reducing waste generation, maximizing atom economy, and employing biocatalysis. Embracing green chemistry principles not only benefits the environment but can also lower production costs and improve process safety.
Common techniques include High-Performance Liquid Chromatography (HPLC) for purity analysis, Nuclear Magnetic Resonance (NMR) spectroscopy for structural identification, Mass Spectrometry (MS) for molecular weight determination, and Infrared (IR) spectroscopy for functional group analysis. These methods ensure the intermediate’s identity and quality.
Strict adherence to Good Manufacturing Practices (GMP) is paramount. Regulatory agencies like the FDA and EMA require detailed documentation of manufacturing processes, quality control procedures, and impurity profiles. Compliance ensures the safety, efficacy, and consistency of the final drug product and is essential for market access. Regular audits and inspections are conducted to verify compliance.
Continuous flow chemistry offers numerous advantages, including improved process control, enhanced safety, increased efficiency, and reduced waste. It allows for precise control of reaction parameters and enables scalability. Its inherently safer nature also makes it attractive for handling hazardous reagents and reactions. This technology is becoming increasingly prevalent in drug intermediate production.
In conclusion, drug intermediates list are the unsung heroes of the pharmaceutical industry, playing a critical role in ensuring the availability of life-saving medications. Understanding their importance, the key factors influencing their quality and cost, and the emerging trends shaping their future is crucial for success in this dynamic field. From navigating complex supply chains to embracing sustainable manufacturing practices, pharmaceutical companies must prioritize innovation and collaboration to overcome current challenges and capitalize on future opportunities.
Looking ahead, the adoption of advanced technologies like AI, blockchain, and continuous flow chemistry will revolutionize the drug intermediates list landscape. Investing in skilled workforce development and fostering strong partnerships will be essential for driving innovation and ensuring a resilient and sustainable supply chain. To learn more about our comprehensive range of drug intermediates list and how we can support your pharmaceutical manufacturing needs, visit our website today: drug intermediates list.