Pharmaceutical intermediates sourcing is a critical component of the global pharmaceutical supply chain, playing a vital role in the production of life-saving medications. This process involves the procurement of chemical compounds – the building blocks for active pharmaceutical ingredients (APIs) – from various suppliers worldwide. Efficient and reliable pharmaceutical intermediates sourcing directly impacts drug availability, cost, and quality, making it a subject of increasing importance for pharmaceutical companies, healthcare providers, and patients globally.
The demand for pharmaceutical intermediates is consistently rising, driven by an aging global population, increasing prevalence of chronic diseases, and continuous innovation in drug development. However, this demand is met with complexities, including geopolitical risks, regulatory hurdles, and the need for stringent quality control. Effective pharmaceutical intermediates sourcing strategies are therefore essential for mitigating these challenges and ensuring a stable and secure drug supply.
Understanding the intricacies of pharmaceutical intermediates sourcing allows organizations to optimize their supply chains, reduce costs, and maintain the highest standards of product quality. It also fosters a more resilient and responsive pharmaceutical industry, capable of addressing emerging health crises and meeting the evolving needs of patients worldwide. Specialized companies like pharmaceutical intermediates sourcing offer expertise in navigating this complex landscape.

The global pharmaceutical industry is a multi-trillion dollar market, deeply reliant on a complex network of suppliers and manufacturers. Pharmaceutical intermediates sourcing forms the foundational layer of this network, representing a significant portion of the overall cost structure and influencing the speed and agility of drug production. According to the United Nations Comtrade database, the trade value of organic chemicals – a key category encompassing many pharmaceutical intermediates – exceeded $350 billion in 2022, highlighting the scale of this global market.
The increasing globalization of pharmaceutical manufacturing has amplified the need for robust and diversified sourcing strategies. While historically concentrated in developed nations, the production of pharmaceutical intermediates is now heavily concentrated in regions like India and China, offering cost advantages but also introducing geopolitical and supply chain vulnerabilities. The COVID-19 pandemic dramatically illustrated these vulnerabilities, disrupting supply chains and highlighting the critical importance of resilient pharmaceutical intermediates sourcing.
The challenge lies in balancing cost efficiency with security of supply, quality assurance, and adherence to increasingly stringent regulatory requirements. Pharmaceutical companies are actively seeking to optimize their sourcing practices through strategies like dual sourcing, regional diversification, and closer collaboration with suppliers, all aimed at mitigating risks and ensuring a consistent flow of essential intermediates. Companies specializing in pharmaceutical intermediates sourcing play a crucial role in this optimization.
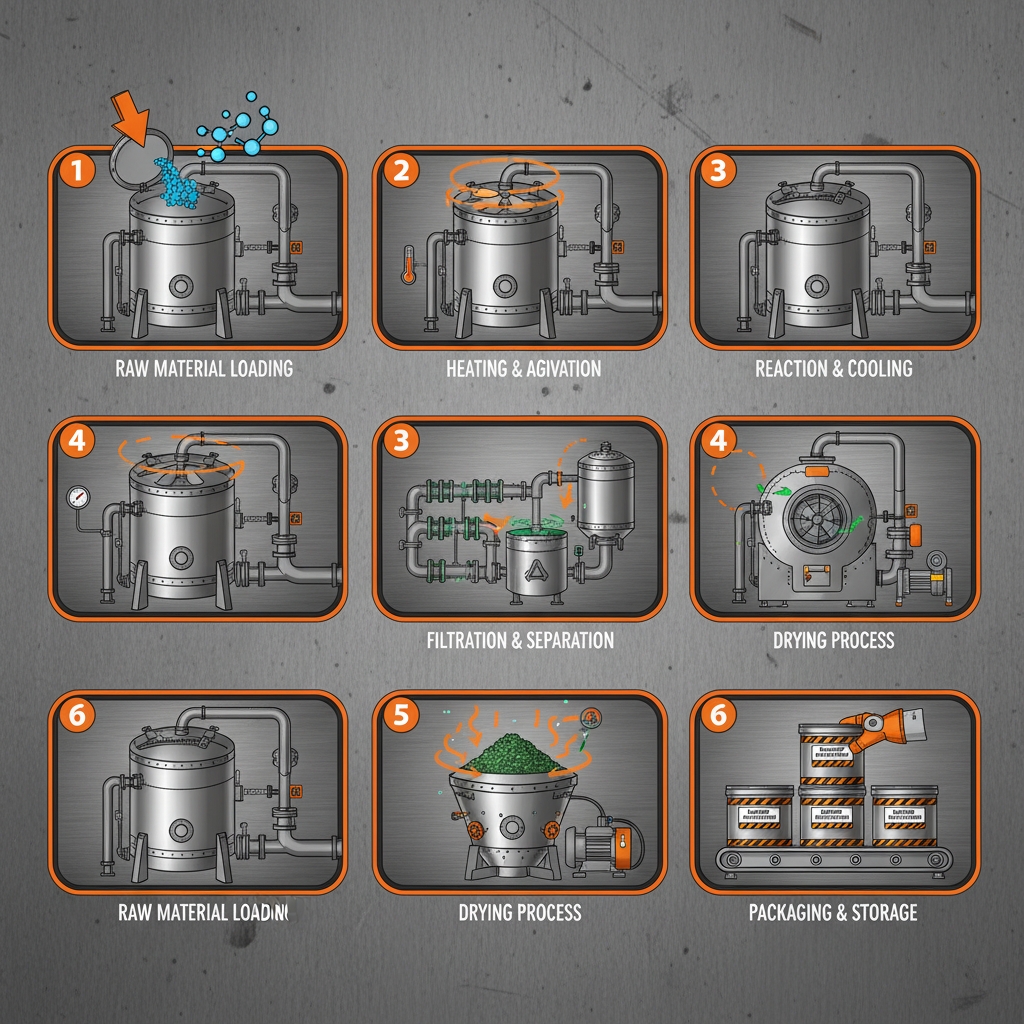
Pharmaceutical intermediates sourcing refers to the process of identifying, evaluating, and procuring chemical compounds that are used as raw materials in the synthesis of active pharmaceutical ingredients (APIs). These intermediates are not the final drug product but are essential building blocks that undergo further chemical transformations to create the API. The sourcing process encompasses everything from supplier qualification and contract negotiation to quality control, logistics, and regulatory compliance.
This practice is fundamental to modern pharmaceutical manufacturing because synthesizing APIs from basic raw materials is often complex, costly, and time-consuming. Utilizing pre-made intermediates streamlines the production process, reduces manufacturing time, and lowers overall costs. In essence, it allows pharmaceutical companies to focus their resources on the final stages of drug development and formulation, rather than reinventing the wheel for every API they produce.
The importance of pharmaceutical intermediates sourcing extends beyond purely commercial considerations. Access to affordable and reliable APIs is critical for ensuring global healthcare equity, particularly in developing countries where access to essential medicines is often limited. Responsible pharmaceutical intermediates sourcing practices also contribute to sustainable manufacturing and reduce the environmental impact of the pharmaceutical industry.
Several core components underpin successful pharmaceutical intermediates sourcing. Quality Assurance is paramount, requiring rigorous testing, adherence to Good Manufacturing Practices (GMP), and comprehensive documentation to ensure the purity, potency, and safety of the sourced materials. Establishing robust quality control procedures is non-negotiable in this industry.
Cost Efficiency is another critical factor, but it must be balanced with quality and reliability. Sourcing from low-cost regions can offer significant savings, but it's crucial to avoid compromising on quality or increasing supply chain risks. A holistic cost analysis, considering factors beyond just the purchase price, is essential. Partnering with a skilled pharmaceutical intermediates sourcing firm can often help optimize these costs.

Pharmaceutical intermediates sourcing is universally applied across the pharmaceutical industry, regardless of the specific therapeutic area or geographical location. Major pharmaceutical hubs, such as the United States, Europe, and Japan, rely heavily on sourced intermediates for the production of a vast array of drugs, from generic medications to innovative biologics. In India, a major pharmaceutical manufacturing center, local companies actively source intermediates both domestically and internationally to fuel their production capabilities.
The application extends beyond large-scale commercial manufacturing. Research and development organizations, including universities and biotechnology companies, frequently require specialized intermediates for drug discovery and pre-clinical studies. Custom synthesis and sourcing services cater to these niche demands, providing access to compounds that may not be commercially available. In post-disaster relief operations, the rapid availability of essential medicines often hinges on the efficient sourcing of intermediates to quickly ramp up API production.
Furthermore, in remote industrial zones where local chemical manufacturing infrastructure is limited, pharmaceutical intermediates sourcing provides a vital link to the global supply chain, enabling the production of essential medicines closer to the point of need. This is particularly crucial in regions with limited access to healthcare and a high burden of disease.
The advantages of strategic pharmaceutical intermediates sourcing are multifaceted. From a financial perspective, it can lead to significant cost savings through optimized procurement and reduced manufacturing expenses. However, the long-term value extends far beyond mere cost reduction. A robust sourcing strategy fosters a more resilient supply chain, mitigating risks associated with geopolitical instability, natural disasters, or supplier disruptions.
Sustainability is another key benefit. By partnering with suppliers committed to responsible manufacturing practices and environmental stewardship, pharmaceutical companies can reduce their carbon footprint and contribute to a more sustainable pharmaceutical industry. This enhances their corporate social responsibility profile and builds trust with stakeholders. A reliable pharmaceutical intermediates sourcing partner can guide companies through these considerations.
Ultimately, effective pharmaceutical intermediates sourcing directly translates to improved patient outcomes. By ensuring the timely availability of high-quality APIs, it contributes to the consistent production of life-saving medications, enhancing access to healthcare and improving the overall quality of life for patients worldwide. It's a critical component of a trustworthy and ethical pharmaceutical industry.
The future of pharmaceutical intermediates sourcing is poised for significant transformation, driven by technological advancements and evolving industry priorities. Digitalization, including the adoption of blockchain technology, is gaining momentum, offering enhanced traceability and transparency throughout the supply chain. This can help combat counterfeit products and ensure the authenticity of sourced materials.
Green chemistry and sustainable manufacturing practices are also becoming increasingly important. The development of more environmentally friendly synthesis routes and the utilization of renewable feedstocks will reduce the environmental impact of pharmaceutical intermediate production. Automation and artificial intelligence (AI) are expected to play a greater role in optimizing sourcing decisions and improving supply chain efficiency.
Despite advancements, pharmaceutical intermediates sourcing continues to face challenges. Supply chain disruptions, often stemming from geopolitical events or natural disasters, remain a constant threat. Increasing regulatory scrutiny and the need for enhanced quality control add complexity to the process. Furthermore, the rise of counterfeit products poses a significant risk to patient safety.
Solutions include diversifying sourcing locations to reduce reliance on single suppliers, implementing robust risk management strategies, and investing in advanced analytical technologies for quality control. Collaboration and information sharing among industry stakeholders are also crucial. Leveraging the expertise of specialized pharmaceutical intermediates sourcing companies can provide access to best practices and mitigate potential risks.
Moreover, adopting a proactive approach to regulatory compliance and embracing digitalization can streamline processes and improve transparency, ultimately building a more resilient and secure pharmaceutical supply chain.
| Challenge Area | Severity (1-10) | Impact on Cost | Mitigation Strategy |
|---|---|---|---|
| Geopolitical Instability | 8 | High | Diversify Sourcing, Regional Stockpiles |
| Quality Control Issues | 9 | Medium | Robust Auditing, Advanced Analytics |
| Regulatory Compliance | 7 | Medium | Dedicated Compliance Team, SOPs |
| Counterfeit Products | 6 | High | Blockchain Traceability, Secure Packaging |
| Supply Chain Transparency | 5 | Low | Digitalization, Supplier Mapping |
| Lead Times & Delays | 7 | Medium | Strategic Inventory, Alternative Suppliers |
China generally offers larger production capacities and a wider range of intermediates, often at highly competitive prices. However, India is increasingly known for its strong API manufacturing capabilities and a more robust regulatory framework. Choosing between the two depends on specific needs, risk tolerance, and quality requirements. Careful due diligence and supplier qualification are essential in both cases.
Establishing a comprehensive quality assurance program is crucial. This includes thorough supplier audits, rigorous testing of incoming materials, adherence to Good Manufacturing Practices (GMP), and comprehensive documentation. Utilizing analytical techniques like HPLC, GC-MS, and NMR spectroscopy can verify purity and identify potential contaminants. Partnering with reputable pharmaceutical intermediates sourcing providers can also ensure consistent quality.
Regulatory compliance is paramount. Pharmaceutical intermediates must meet the standards set by regulatory bodies like the FDA (US), EMA (Europe), and PMDA (Japan). This includes providing Certificates of Analysis (CoAs), ensuring compliance with REACH regulations, and maintaining proper documentation throughout the supply chain. Failure to comply can result in significant penalties and product recalls.
Diversifying sourcing locations, establishing buffer stocks, and developing strong relationships with multiple suppliers are key strategies. Conducting thorough risk assessments, implementing contingency plans, and continuously monitoring the geopolitical landscape can also help mitigate potential disruptions. Proactive risk management is crucial for ensuring a stable and reliable supply of intermediates.
Sustainability is increasingly important. Pharmaceutical companies are actively seeking suppliers who prioritize environmentally friendly manufacturing processes, reduce waste generation, and minimize their carbon footprint. The use of green chemistry principles and renewable feedstocks is gaining traction, contributing to a more sustainable pharmaceutical industry.
Digitalization, including the use of blockchain technology, can enhance traceability, transparency, and efficiency in the supply chain. AI-powered analytics can optimize sourcing decisions, predict potential disruptions, and improve inventory management. Digital platforms facilitate better communication and collaboration among stakeholders, streamlining the entire process.
In conclusion, pharmaceutical intermediates sourcing is a complex but essential function within the global pharmaceutical industry. Its success hinges on a careful balance of cost efficiency, quality assurance, regulatory compliance, and risk management. By embracing innovation, prioritizing sustainability, and fostering collaboration, organizations can build more resilient, transparent, and efficient supply chains. Understanding the intricacies of this field is no longer just a strategic advantage, but a necessity for ensuring the availability of safe and effective medicines worldwide.
Looking ahead, the future of pharmaceutical intermediates sourcing will be shaped by technological advancements and evolving industry priorities. Investing in digitalization, adopting green chemistry principles, and fostering stronger supplier relationships will be critical for navigating the challenges and capitalizing on the opportunities that lie ahead. We encourage you to explore further resources and consider partnering with experts like pharmaceutical intermediates sourcing to optimize your sourcing strategies.