Intermediate pharma, the production of chemical compounds used as building blocks for pharmaceuticals, plays a critical role in the global healthcare supply chain. Its importance stems from the complex and specialized nature of drug manufacturing, where sourcing high-quality, consistent intermediates is paramount. These compounds represent a significant value proposition, offering cost-effective routes to final drug products and contributing substantially to the affordability and accessibility of medications worldwide.
The global intermediate pharma market is experiencing substantial growth, driven by factors like increasing demand for generic drugs, the rise of contract manufacturing organizations (CMOs), and the growing prevalence of chronic diseases. According to a report by the United Nations Conference on Trade and Development (UNCTAD), pharmaceutical trade, including intermediates, reached over $300 billion in 2022, demonstrating the sector’s significant economic impact. However, this growth also presents challenges, including ensuring supply chain resilience, adhering to stringent regulatory standards, and mitigating environmental concerns.
Understanding intermediate pharma is vital for stakeholders across the pharmaceutical industry—from research and development teams and manufacturing facilities to regulatory bodies and healthcare providers. A clear grasp of the processes, challenges, and future trends within this sector is crucial for maintaining a stable and efficient drug supply, ultimately impacting public health outcomes. Companies like intermediate pharma are at the forefront of addressing these critical needs.


Intermediate pharma forms the backbone of modern pharmaceutical production. Without consistent access to these crucial building blocks, the manufacture of vital medications would be severely hampered. The complexity of drug synthesis often necessitates multiple intermediate steps, each requiring precise control and rigorous quality assurance. This intricate process underpins the ability to produce both innovative and generic drugs, fulfilling global healthcare needs.
The demand for intermediate pharma is closely tied to advancements in pharmaceutical research and the increasing prevalence of chronic diseases. As new therapeutic targets are identified and drug pipelines expand, the need for specialized intermediates grows correspondingly. The reliable sourcing of these compounds is therefore critical for ensuring continued innovation and the ongoing availability of life-saving treatments.
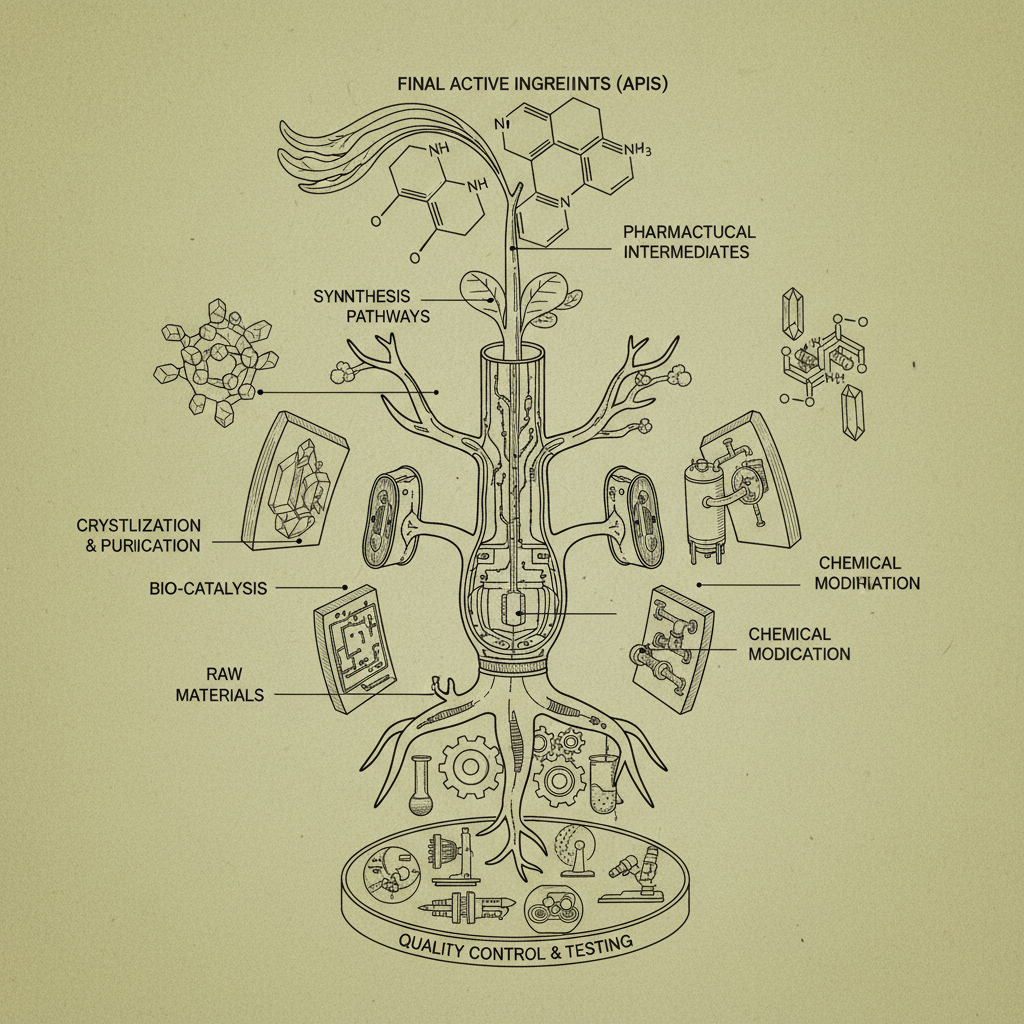
Intermediate pharma refers to the chemical compounds that are synthesized during the multi-step process of creating an Active Pharmaceutical Ingredient (API). These intermediates are not the final drug substance itself, but rather precursors that undergo further chemical transformations to achieve the desired API structure. They represent a critical stage in the drug manufacturing process, requiring specialized expertise and adherence to strict quality control measures.
The connection to modern industry stems from the increasing complexity of pharmaceutical compounds. Most drugs are not created in a single step; instead, they require a sequence of carefully controlled reactions to build the desired molecular structure. This reliance on multi-step synthesis necessitates the production and purification of various intermediate compounds. intermediate pharma companies specialize in this crucial area, offering customized synthesis and large-scale production capabilities.
Understanding intermediate pharma is also essential for humanitarian needs. Ensuring a stable and affordable supply of essential medicines requires efficient and reliable intermediate production. Disruptions in the intermediate supply chain can lead to drug shortages, impacting vulnerable populations and hindering global health initiatives.
Several factors critically influence the quality of intermediate pharma. Purity is paramount; even trace impurities can impact the final API’s efficacy and safety. Precise control of reaction conditions, meticulous purification techniques, and rigorous analytical testing are essential for achieving the required purity levels. Consistent quality across batches is also vital, ensuring reproducibility in the drug manufacturing process.
Regulatory compliance is another crucial factor. Intermediate pharma production must adhere to Good Manufacturing Practices (GMP) guidelines, ensuring that the processes are well-defined, controlled, and documented. This includes maintaining detailed records of all materials used, equipment calibration, and analytical results. Compliance with regulations from bodies like the FDA and EMA is non-negotiable for market access.
Finally, supply chain transparency and traceability are becoming increasingly important. Knowing the origin of raw materials, tracking the intermediate through each stage of production, and ensuring proper documentation are essential for building trust and mitigating risks.
Effective quality control systems, including in-process monitoring and final product testing, are vital for verifying that the intermediate meets the specified quality attributes. This often involves advanced analytical techniques like HPLC, NMR, and mass spectrometry.
Intermediate pharma finds applications across a wide spectrum of pharmaceutical sectors, including small molecule drugs, biologics, and generic pharmaceuticals. In the production of small molecule drugs, intermediates often represent the majority of the synthetic steps, demanding intricate chemical transformations and purification processes.
The generic drug industry heavily relies on sourcing high-quality intermediates to reduce production costs and maintain competitive pricing. CMOs (Contract Manufacturing Organizations) also utilize intermediate pharma extensively, providing customized synthesis services to pharmaceutical companies. intermediate pharma expertise allows them to efficiently manufacture APIs to client specifications.
Utilizing specialized intermediate pharma suppliers offers significant cost advantages. Internal API manufacturing often requires substantial capital investment in equipment and personnel. Outsourcing intermediate production allows pharmaceutical companies to reduce these overhead costs and focus on core competencies.
Furthermore, a diversified supply chain built on reliable intermediate sources enhances supply chain security. Relying on a single API supplier can create vulnerabilities to disruptions. Access to multiple intermediate sources mitigates these risks, ensuring a consistent supply of critical drug components.
The future of intermediate pharma is being shaped by two major trends: green chemistry and digitalization. Green chemistry focuses on developing more sustainable and environmentally friendly synthetic routes, minimizing waste, and utilizing renewable resources. This includes adopting biocatalysis, flow chemistry, and alternative solvents.
Digitalization, including the implementation of advanced process analytical technologies (PAT) and artificial intelligence (AI), is revolutionizing intermediate pharma manufacturing. AI algorithms can optimize reaction conditions, predict product quality, and automate quality control processes, leading to increased efficiency and reduced costs.
One significant challenge is the increasing regulatory scrutiny surrounding impurities and residual solvents. Meeting stringent regulatory requirements necessitates advanced purification techniques and analytical capabilities.
Another challenge is the complexity of synthesizing chiral intermediates, which are essential for many pharmaceutical compounds. Solutions include employing chiral catalysts, resolving racemic mixtures, and utilizing asymmetric synthesis methods. intermediate pharma companies are investing in these advanced technologies to address these challenges.
Finally, maintaining supply chain resilience in the face of geopolitical uncertainties and natural disasters is crucial. Building strategic partnerships with multiple suppliers, diversifying sourcing locations, and implementing robust inventory management systems can mitigate these risks.
| Purity Level (%) | Yield Rate (%) | Production Cost ($/kg) | Regulatory Compliance Score (1-10) |
|---|---|---|---|
| 99.5 | 85 | 500 | 9 |
| 99.8 | 90 | 400 | 10 |
| 99.2 | 78 | 600 | 7 |
| 99.7 | 88 | 450 | 9 |
| 99.4 | 82 | 550 | 8 |
| 99.6 | 86 | 480 | 10 |
Lead times for custom synthesis vary significantly depending on the complexity of the molecule, the quantity required, and current production capacity. Generally, a straightforward, well-documented synthesis of a gram-scale intermediate can take 2-4 weeks, while more complex, multi-step syntheses at larger scales may require several months. Factors such as reagent availability and purification challenges can also influence the timeline.
We employ a rigorous quality control system that encompasses multiple stages, beginning with raw material testing and continuing through in-process monitoring and final product analysis. Techniques like HPLC, NMR, and mass spectrometry are used to verify purity and identity. All processes adhere to strict GMP guidelines, and comprehensive documentation is maintained throughout the manufacturing process.
We provide a comprehensive documentation package that typically includes a Certificate of Analysis (CoA), a Material Safety Data Sheet (MSDS), and, upon request, detailed manufacturing process information and analytical data. We can also provide documentation to support regulatory filings, such as DMFs (Drug Master Files), as needed.
Our MOQ varies based on the complexity of the synthesis and the specific requirements of the project. We can often accommodate smaller quantities for initial development and optimization phases. However, larger-scale production typically requires a higher MOQ to ensure cost-effectiveness. We are happy to discuss specific project needs and provide a tailored quote.
Yes, we offer comprehensive process development and optimization services. Our team of experienced chemists can work with you to develop new synthetic routes, optimize existing processes for improved yield and purity, and scale up production from laboratory scale to commercial quantities.
We understand the importance of protecting our clients’ confidential information and intellectual property. We have strict confidentiality agreements in place, and our facilities are secured to prevent unauthorized access. All personnel are trained on confidentiality protocols, and we are committed to maintaining the highest level of data security.
In conclusion, intermediate pharma represents a vital link in the global pharmaceutical supply chain. Its importance extends beyond simply providing building blocks for drugs; it encompasses cost-effectiveness, supply chain security, and the potential for sustainable manufacturing practices. The ongoing advancements in green chemistry, digitalization, and process optimization are poised to further enhance the efficiency and reliability of intermediate pharma production, ultimately benefiting patients worldwide.
As the pharmaceutical landscape continues to evolve, a strategic approach to intermediate sourcing and manufacturing will be critical for success. Companies like intermediate pharma, dedicated to innovation and quality, will play an increasingly crucial role in shaping the future of drug development and ensuring access to life-saving medications.