- Afrikaans
- Albanian
- Amharic
- Arabic
- Armenian
- Azerbaijani
- Basque
- Belarusian
- Bengali
- Bosnian
- Bulgarian
- Catalan
- Cebuano
- Corsican
- Croatian
- Czech
- Danish
- Dutch
- English
- Esperanto
- Estonian
- Finnish
- French
- Frisian
- Galician
- Georgian
- German
- Greek
- Gujarati
- Haitian Creole
- hausa
- hawaiian
- Hebrew
- Hindi
- Miao
- Hungarian
- Icelandic
- igbo
- Indonesian
- irish
- Italian
- Japanese
- Javanese
- Kannada
- kazakh
- Khmer
- Rwandese
- Korean
- Kurdish
- Kyrgyz
- Lao
- Latin
- Latvian
- Lithuanian
- Luxembourgish
- Macedonian
- Malgashi
- Malay
- Malayalam
- Maltese
- Maori
- Marathi
- Mongolian
- Myanmar
- Nepali
- Norwegian
- Norwegian
- Occitan
- Pashto
- Persian
- Polish
- Portuguese
- Punjabi
- Romanian
- Russian
- Samoan
- Scottish Gaelic
- Serbian
- Sesotho
- Shona
- Sindhi
- Sinhala
- Slovak
- Slovenian
- Somali
- Spanish
- Sundanese
- Swahili
- Swedish
- Tagalog
- Tajik
- Tamil
- Tatar
- Telugu
- Thai
- Turkish
- Turkmen
- Ukrainian
- Urdu
- Uighur
- Uzbek
- Vietnamese
- Welsh
- Bantu
- Yiddish
- Yoruba
- Zulu
LIHLAHISO KOPO
Re lula re hlahisa lihlahisoa tse ncha ho khothaletsa nts'etsopele ea moshoelella.
Mabapi le Khampani
Re lula re hlahisa lihlahisoa tse ncha ho khothaletsa nts'etsopele ea moshoelella.
Boemo ba Khoebo
Shijiazhuang Kunxiangda Technology Co., Ltd.
e thehiloe ka 2011. re setsebi sa ho etsa li-intermediates tsa meriana le k'hamphani ea khoebo bakeng sa li-intermediates tsa dae, le lisebelisoa tse ling tsa lik'hemik'hale tse tala. Feme ea rona e sebakeng sa moruo le nts'etsopele ea theknoloji ea toropo ea Shijiazhuang, profinseng ea Hebei. Kakaretso ea libaka e ka ba lihekthere tse 50, 'me ho na le basebetsi ba fetang 300, ho kenyeletsoa le basebetsi ba 9.
-
0
Lilemo tsa Boiphihlelo
-
0
Linaha tse Romellang
-
0
Basebetsi ba Hona Joale

Mmaraka oa rona oa Lefatše
Lihlahisoa li romelloa Europe, Amerika Boroa, Asia, Middle East le linaha le libaka tse ling tse ngata, hape li thehile marang-rang a holileng le a tsitsitseng a ho bapatsa.
6Likontinente
35Linaha kapa Libaka
Bala Litaba Tsa Rōna Tsa Morao-rao
Re lula re hlahisa lihlahisoa tse ncha ho khothaletsa nts'etsopele ea moshoelella.

Apr.16,2026
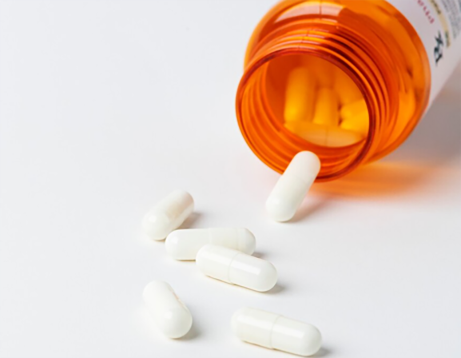
Strategic Growth and Quality Guide for api pharma business
The global pharmaceutical landscape is evolving rapidly, placing a heightened emphasis on the quality and reliability of Active Pharmaceutical Ingredients (APIs). Navigating the api pharma business requires a deep understanding of regulatory compliance, chemical synthesis innovation, and supply chain resilience. For companies aiming to scale, the ability to source high-purity intermediates and maintain stringent quality control is the cornerstone of success. In this guide, we will explore the critical components of managing a successful API enterprise, from regulatory hurdles to the integration of green chemistry, ensuring your business remains competitive in an increasingly demanding market.
Bala Litaba Tse Ling
Apr.09,2026
Importance and Quality Standards of api of pharmaceutical
In the complex world of drug manufacturing, the api of pharmaceutical, or Active Pharmaceutical Ingredient, serves as the biological heart of any medication. While a pill may contain various fillers, binders, and coatings, the API is the specific component responsible for producing the desired therapeutic effect in the patient. Ensuring the purity, stability, and efficacy of these ingredients is not just a technical requirement but a critical safety mandate for global healthcare. From simple pain relievers to complex oncology treatments, the quality of the API dictates the success of the final medical product.
Bala Litaba Tse Ling
Apr.07,2026
Comprehensive Guide to API of Drugs Global Sourcing and Quality Control
In the complex world of pharmaceutical ingredient sourcing, understanding the API of drugs – Active Pharmaceutical Ingredients – is paramount. These are the biologically active components that produce the intended effects in medications. This article will provide a detailed overview of APIs, their sourcing, quality control, and the role of trusted suppliers like KXD Chem in navigating the global market. Effective API sourcing is crucial for pharmaceutical companies aiming for cost-efficiency, quality, and regulatory compliance.
Bala Litaba Tse Ling